Ley, D.; Gerbig, D.; Schreiner, P. R. Chem. Sci. 2013, 4, 677
Contributed by Steven Bachrach.
Reposted from Computational Organic Chemistry with permission

This work is licensed under a Creative Commons Attribution-NoDerivs 3.0 Unported License.
Contributed by Steven Bachrach.
Reposted from Computational Organic Chemistry with permission
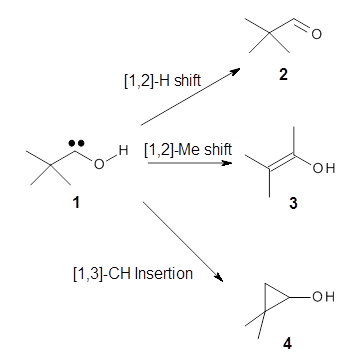
Sorry I missed this paper from much earlier this year – it’s from a journal that’s not on my normal reading list. Anyways, here is another fantastic work from the Schreiner lab demonstrating the concept of tunneling control (see this post).1 They prepare the t-butylhydroxycarbene 1 at low temperature to look for evidence of formation of possible products arising from a [1,2]-hydrogen shift (2), a [1,2]-methyl shift (3) or a [1,3]-CH insertion (4).

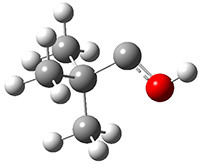
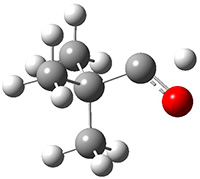
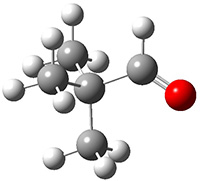
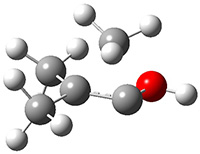
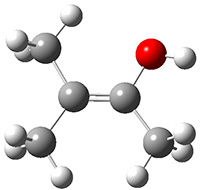
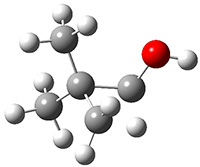
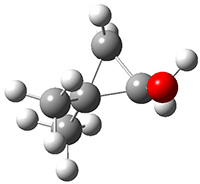
Schreiner performed CCSD(T)/cc-pVDZ optimizations of these compounds along with the transition states for the three migrations. The optimized geometries and relative energies are shown in Figure 1. The thermodynamic product is the aldehyde 2 while the kinetic product is the cyclopropane 4, with a barrier of 23.8 kcal mol-1 some 3.5 kcal mol-1 lower than the barrier leading to 2.
1
(0.0) |
TS2
(27.3) |
2
(-53.5) |
TS3
(31.0) |
3
(-41.0) | |
TS4
(23.8) |
4
(-28.3) |
Figure 1. CCSD(T)/cc-pVDZ optimized structures of 1-4 and the transition states for the three reaction. Relative energies in kcal mol-1.
At low temperature (11 K), 1 is found to slowly convert into 2 with a half-life of 1.7 h. No other product is observed. Rates for the three reactions were also computed using the Wentzel-Kramers-Brillouin (WKB) method (which Schreiner and Allen have used in all of their previous studies). The predicted rate for the conversion of 1 into 2, which takes place at 11 K solely through a tunneling process, is 0.4h, in quite reasonable agreement with experiment. The predicted rates for the other two potential reactions at 11 K are 1031 and 1040 years.
This is clearly an example of tunneling control. The reaction occurs not across the lowest barrier, butthrough the narrowest barrier.
References
(1) Ley, D.; Gerbig, D.; Schreiner, P. R. "Tunneling control of chemical reactions: C-H insertion versus H-tunneling in tert-butylhydroxycarbene," Chem. Sci. 2013, 4, 677-684, DOI: 10.1039/C2SC21555A.
InChI
1: InChI=1S/C5H10O/c1-5(2,3)4-6/h6H,1-3H3
InChIKey=ZGFKBRGJTPEEOC-UHFFFAOYSA-N
InChIKey=ZGFKBRGJTPEEOC-UHFFFAOYSA-N
2: InChI=1S/C5H10O/c1-5(2,3)4-6/h4H,1-3H3
3: InChI=1S/C5H10O/c1-4(2)5(3)6/h6H,1-3H3
InChIKey=BZAZNULYLRVMSW-UHFFFAOYSA-N
InChIKey=BZAZNULYLRVMSW-UHFFFAOYSA-N
4: InChI=1S/C5H10O/c1-5(2)3-4(5)6/h4,6H,3H2,1-2H3
InChIKey=MWWQKEGWQLBJBJ-UHFFFAOYSA-N
InChIKey=MWWQKEGWQLBJBJ-UHFFFAOYSA-N

This work is licensed under a Creative Commons Attribution-NoDerivs 3.0 Unported License.
No comments:
Post a Comment