Toriumi, N.; Muranaka, A.; Hirano, K.; Yoshida, K.; Hashizume, D.; Uchiyama, M. Angew. Chem. Int. Ed. 2014, 53, 7814-7818
Contributed by Steven Bachrach.
Reposted from Computational Organic Chemistry with permission

This work is licensed under a Creative Commons Attribution-NoDerivs 3.0 Unported License.
Contributed by Steven Bachrach.
Reposted from Computational Organic Chemistry with permission
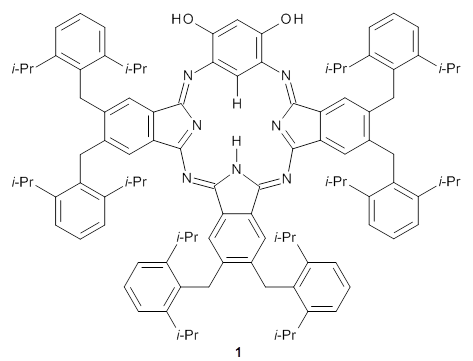
Muranaka and Uchiyama have prepared an 18π-electron system that exhibits variable aromaticity in its tautomeric forms.1 The synthesized benziphthalacyanine 1 shows upfield resonances in the 1H NMR for the internal hydrogens: 1.89 ppm for the C-H proton and 4.67 ppm for the N-H proton. This indicates some weak diatropicity.

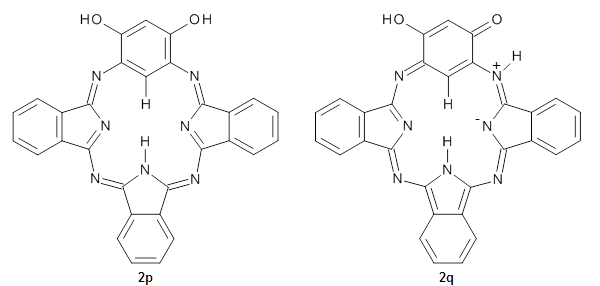
To address this interesting magnetic property, they reported B3LYP/6-31+G(d) computations on the model system 2 in its phenol 2p and quinoidal 2q tautomeric forms.

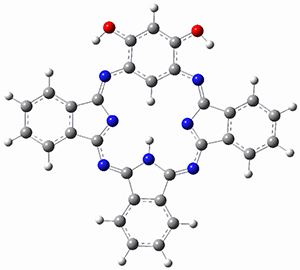
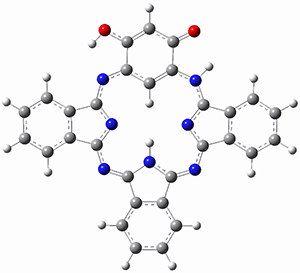
The optimized structures are shown in Figure 1. The phenol form 2p has NICS(0) and NICS(1) values of -6.77 and -6.04 ppm, respectively, indicating only modest aromaticity. However, the NICS values for the quinoidal from 2q are much more negative, -11.43 (NICS(0)) and -10.10 (NICS(1)) ppm, indicating a more significant aromatic character. The calculated chemical shift of the internal C-H is most telling: for 2q it is -4.55ppm but for 2p it is 0.97 ppm, in good agreement with experiment. Thus, 1 has an 18π-electron modestly aromatic periphery, with the phenol form dominant. There is no evidence of a 20π-electron periphery.
2p
|
2q
|
Figure 1. B3LYP/6-31+G(d) optimized geometries of 2p and 2q.
(Note that the supporting materials have a missing carbon in 2q and I have made a guess at its proper location – so this is not quite the optimized structure! Once again, a statement about the quality of SI!)
References
(1) Toriumi, N.; Muranaka, A.; Hirano, K.; Yoshida, K.; Hashizume, D.; Uchiyama, M. "18π-Electron Tautomeric Benziphthalocyanine: A Functional Near-Infrared Dye with Tunable Aromaticity," Angew. Chem. Int. Ed. 2014, 53, 7814-7818, DOI: 10.1002/anie.201404020.
InChIs
1: InChI=1S/C108H125N7O2/c1-57(2)75-31-25-32-76(58(3)4)87(75)43-69-49-93-95(51-71(69)45-89-79(61(9)10)35-27-36-80(89)62(11)12)105-111-103(93)109-99-55-100(102(117)56-101(99)116)110-104-94-50-70(44-88-77(59(5)6)33-26-34-78(88)60(7)8)72(46-90-81(63(13)14)37-28-38-82(90)64(15)16)52-96(94)106(112-104)114-108-98-54-74(48-92-85(67(21)22)41-30-42-86(92)68(23)24)73(53-97(98)107(113-105)115-108)47-91-83(65(17)18)39-29-40-84(91)66(19)20/h25-42,49-68,116-117H,43-48H2,1-24H3,(H,109,110,111,112,113,114,115)
InChIKey=LCYQUXHUTZWPDZ-UHFFFAOYSA-N
InChIKey=LCYQUXHUTZWPDZ-UHFFFAOYSA-N
2p: InChI=1S/C30H17N7O2/c38-23-14-24(39)22-13-21(23)31-25-15-7-1-3-9-17(15)27(33-25)35-29-19-11-5-6-12-20(19)30(37-29)36-28-18-10-4-2-8-16(18)26(32-22)34-28/h1-14,38-39H,(H,31,32,33,34,35,36,37)
InChIKey=JBKUPBCBFUTSRM-UHFFFAOYSA-N
InChIKey=JBKUPBCBFUTSRM-UHFFFAOYSA-N
2q: InChI=1S/C30H17N7O2/c38-23-14-24(39)22-13-21(23)31-25-15-7-1-3-9-17(15)27(33-25)35-29-19-11-5-6-12-20(19)30(37-29)36-28-18-10-4-2-8-16(18)26(32-22)34-28/h1-14H,(H3,31,32,33,34,35,36,37,38,39)
InChIKey=PSSSGMKTDQVWLR-UHFFFAOYSA-N
InChIKey=PSSSGMKTDQVWLR-UHFFFAOYSA-N

This work is licensed under a Creative Commons Attribution-NoDerivs 3.0 Unported License.
No comments:
Post a Comment