Zhu, J.; An, K.; Schleyer, P. v. R. Org. Lett. 2013, 15, 2442
Contributed by Steven Bachrach.
Reposted from Computational Organic Chemistry with permission

This work is licensed under a Creative Commons Attribution-NoDerivs 3.0 Unported License.
Contributed by Steven Bachrach.
Reposted from Computational Organic Chemistry with permission
One of the most widely recognized principles within organic chemistry is Hückel’s rule: an aromatic compound possesses 4n+2 π-electrons while an antiaromatic compound possesses 4n π-electrons. Much less well known is Baird’s rule:1 the first excited triplet state will be aromatic if it has 4n π-electrons and antiaromatic if it has 4n+2 π-electrons.2
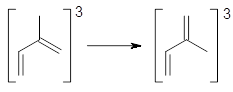
Schleyer used a number of standard methods for assessing aromatic character of a series of excited state triplets, including NICS values and geometric parameters.3 However, Schleyer has long been a proponent of an energetic assessment of aromaticity and it is only now in this recent paper4 that he and co-workers examine the stabilization energy of excited triplet states. The isomerization stabilization energy (ISE)5 compares an aromatic (or antiaromatic) compound against a non-aromatic reference, one that typically is made by appending an exo-methylene group to the ring. So, to assess the ISE of the T1 state of benzene, Reaction 1 is used. (Note that the inherent assumption here is that the stabilization energy of benzene is essentially identical to that of toluene.) At B3LYP/6-311++G(d,p) the energy of Reaction 1 is +13.5 kcal mol-1. This reaction should be corrected for non-conservation of s-cis and s-trans conformers by adding on the energy of Reaction 2, which is +3.4 kcal mol-1. So, the ISE of triplet benzene is +16.9 kcal mol-1, indicating that it is antiaromatic. In contrast, the ISE for triplet cyclooctatetraene is -15.6 kcal mol-1, and when corrected its ISE value is -24.7 kcal mol-1, indicatingaromatic character. These are completely consistent with Baird’s rule. Schleyer also presents an excellent correlation between the computed ISE values for the triplet state of 9 monocyclic polyenes and their NICS(1)zz values.
 |
Reaction 1
|
 |
Reaction 2
|
I want to thank Henrik Ottosson for bringing this paper to my attention and for his excellent seminar on the subject of Baird’s rule on his recent visit to Trinity University.
References
(1) Baird, N. C. "Quantum organic photochemistry. II. Resonance and aromaticity in
the lowest 3ππ* state of cyclic hydrocarbons," J. Am. Chem. Soc. 1972, 94, 4941-4948, DOI:10.1021/ja00769a025.
the lowest 3ππ* state of cyclic hydrocarbons," J. Am. Chem. Soc. 1972, 94, 4941-4948, DOI:10.1021/ja00769a025.
(2) Ottosson, H. "Organic photochemistry: Exciting excited-state aromaticity," Nat Chem 2012, 4, 969-971, DOI: 10.1038/nchem.1518.
(3) Gogonea, V.; Schleyer, P. v. R.; Schreiner, P. R. "Consequences of Triplet Aromaticity in 4nπ-Electron Annulenes: Calculation of Magnetic Shieldings for Open-Shell Species," Angew. Chem. Int. Ed. 1998, 37, 1945-1948, DOI: 10.1002/(SICI)1521-3773(19980803)37:13/14<1945::AID-ANIE1945>3.0.CO;2-E.
(4) Zhu, J.; An, K.; Schleyer, P. v. R. "Evaluation of Triplet Aromaticity by the
Isomerization Stabilization Energy," Org. Lett. 2013, 15, 2442-2445, DOI: 10.1021/ol400908z.
Isomerization Stabilization Energy," Org. Lett. 2013, 15, 2442-2445, DOI: 10.1021/ol400908z.
(5) Schleyer, P. v. R.; Puhlhofer, F. "Recommendations for the Evaluation of Aromatic Stabilization Energies," Org. Lett. 2002, 4, 2873-2876, DOI: 10.1021/ol0261332.

This work is licensed under a Creative Commons Attribution-NoDerivs 3.0 Unported License.
No comments:
Post a Comment