Blümel, M.; Nagasawa, S.; Blackford, K.; Hare, S. R.; Tantillo, D. J.; Sarpong, R., J. Am. Chem. Soc. 2018, 140, 9291-9298
Contributed by Steven Bacharach
Reposted from Computational Organic Chemistry with permission
 '
'
This work is licensed under a Creative Commons Attribution-NoDerivs 3.0 Unported License.
Contributed by Steven Bacharach
Reposted from Computational Organic Chemistry with permission
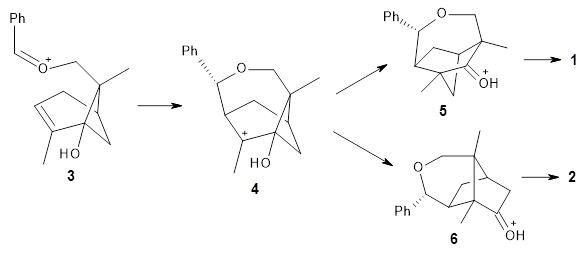
Sarpong and Tantillo have examined the acid-catalyzed Prins/semipinacol rearrangement of hydroxylated pinenes, such as Reaction 1.1
 |
Rxn 1
|
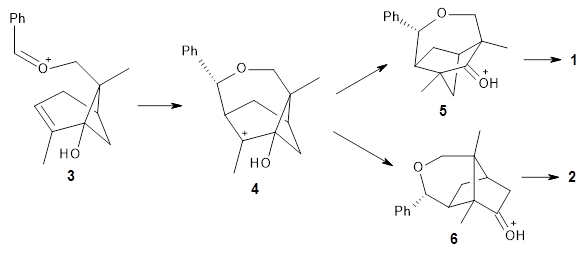
Interestingly, only the fenchone scaffold products, like 1, are observed and the camphor scaffold products, like 2, are not observed. Cation intermediates are likely, and this means that a primary alkyl shift is taking place in preference to a tertiary alkyl shift, see Scheme 1.
Scheme 1.
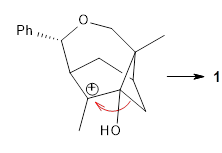 Primary alkyl shift | 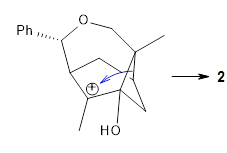 Tertiary alkyl shift |
They proposed the following key steps in the reaction mechanism:

ωB97X-D/6-31+G(d,p) computations find a flat surface around cation intermediate 4: the TS leading to 5and 6 are only 1.3 and 3.3 kcal mol-1, respectively. Since these small barriers are quite susceptible to changes in basis set and functional, and since Tantillo has found many examples of post-transition state bifurcations in cation systems, the authors reasonably decided to conduct molecular dynamics trajectories originating at the TS connecting 3 and 4. The geometries of the critical points are shown in Figure 1.
The trajectory study shows all the usual characteristics of reactions that are under dynamic control. A third of the trajectories show recrossing of the barrier, typical of very flat surfaces. Nearly all of the remaining trajectories led to 5, with only 2 trajectories (~1%) leading to 6. The dynamics are understandable in terms of favoring the primary alkyl shift over the tertiary since a significantly smaller mass needs to move in the former case.
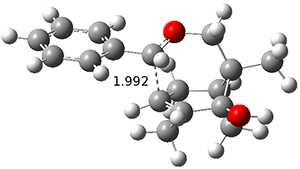 TS 3 → 4 | |
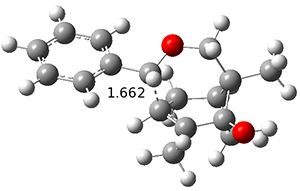 4 | |
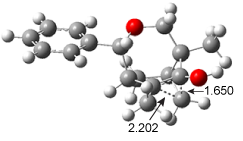 TS 4 → 5 | 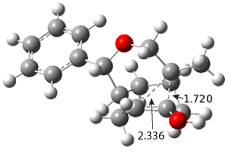 TS 4 → 6 |
Figure 1. ωB97X-D/6-31+G(d,p) optimized geometries.
This is yet another study that implicates dynamic effects in routine reactions, one of many I have discussed over the years.
References
1. Blümel, M.; Nagasawa, S.; Blackford, K.; Hare, S. R.; Tantillo, D. J.; Sarpong, R., "Rearrangement of Hydroxylated Pinene Derivatives to Fenchone-Type Frameworks: Computational Evidence for Dynamically-Controlled Selectivity." J. Am. Chem. Soc. 2018, 140, 9291-9298, DOI: 10.1021/jacs.8b05804.
InChIs
1: InChI=1S/C17H20O2/c1-16-9-12-8-13(16)14(11-6-4-3-5-7-11)19-10-17(12,2)15(16)18/h3-7,12-14H,8-10H2,1-2H3/t12?,13?,14-,16?,17?/m0/s1
InChIKey=LTTUIPPXEHHMJS-XWTIBIIYSA-N
InChIKey=LTTUIPPXEHHMJS-XWTIBIIYSA-N
2: InChI=1S/C17H20O2/c1-16-10-19-15(11-6-4-3-5-7-11)13-8-12(16)9-14(18)17(13,16)2/h3-7,12-13,15H,8-10H2,1-2H3/t12?,13?,15-,16?,17?/m0/s1
InChIKey=GCKIOHNLJYVWKL-CMESGNGWSA-N
InChIKey=GCKIOHNLJYVWKL-CMESGNGWSA-N
 '
'This work is licensed under a Creative Commons Attribution-NoDerivs 3.0 Unported License.
No comments:
Post a Comment