Agnieszka Wołos, Dominik Koszelewski, Rafał Roszak, Sara Szymkuć, Martyna Moskal, Ryszard Ostaszewski, Brenden T. Herrera, Josef M. Maier, Gordon Brezicki, Jonathon Samuel, Justin A. M. Lummiss, D. Tyler McQuade, Luke Rogers & Bartosz A. Grzybowski (2022)
Highlighted by Jan Jensen
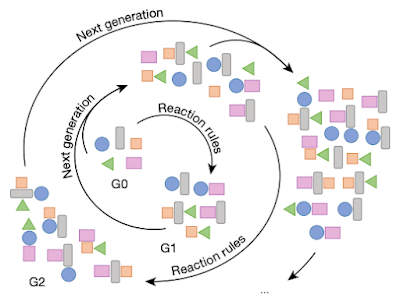
Figure 2a from the paper. (c) 2022 the authors
When I talk to people about retrosynthesis prediction the often mention that synthetic chemists don't tend to use them. There are many reasons for that including various shortcomings of the suggested routes but also the fact that, from a time saving perspective, the retrosynthesis planning makes up a small part of the synthesis process. One common answer to this is "OK, but wait til the robots arrive", but there are several important applications that are applicable right now.
For example, on my own research in de novo molecule discovery I'm often left with hundreds of promising molecules where the only remaining selection criterion is ease of synthesis. Here I routinely use retrosynthesis programs to rank the molecules in terms of number of synthesis steps to make the shortlist of 10-20 molecules that can be presented to experimental collaborators.
This paper presents another example of science that would be impossible without these computational tools. The authors search for reaction networks that connect 189 small molecule waste by-products from chemical industry to 4113 high-value molecules (approved drugs and agrochemicals). The use a reaction prediction algorithm called Allchemy to iteratively generate increasingly complicated molecules and, at each step, bias the search towards the target. Among the 300 million molecules that result from this process the were able to identify 167 target molecules, with an average of 216 synthetic paths per target. The synthetic paths are further ranked using a complicated scoring functions that accounts for all sorts of practical considerations, since aim is to produce large quantities of each target, and a few of the paths are experimentally verified on the kg scale.
One interesting part the approach is the prediction of reaction conditions, which is done in terms of categories: e.g. protic/aprotic and polar/nonpolar solvents, and very low, low, room temperature, high, and very high temperatures. This makes a lot more sense to than trying to predict the exact solvent or temperature.

This work is licensed under a Creative Commons Attribution 4.0 International License.
No comments:
Post a Comment