Cabezas, C.; Varela, M.; Alonso, J. L., Angew. Chem. Int. Ed. 2017, 56, 6420-6425
Contributed by Steven Bacharach
Reposted from Computational Organic Chemistry with permission
 '
'
This work is licensed under a Creative Commons Attribution-NoDerivs 3.0 Unported License.
Contributed by Steven Bacharach
Reposted from Computational Organic Chemistry with permission
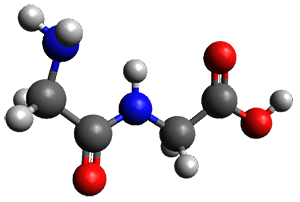
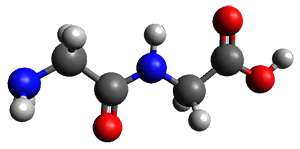
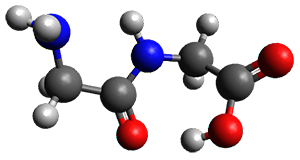
Continuing their application of laser ablation molecular beam Fourier transform microwave (LA-MB-FTMW) spectroscopy and computational chemistry to biochemical molecules (see these previous posts), the Alonso group reports on the structure of the glycine-glycine dipeptide 1.1 The microwave spectrum shows three different conformers. MP2/6-311++G(d,p) computations, the same method they have previously utilized for predicting geometries, revealed a number of different conformations. By matching the spectroscopic parameters obtained from the spectrum with those of the computed structures, they proposed the three conformations 1a, 1b, and 1c, shown in Figure 1.
1a
|
1b
|
1c
|
Figure 1. ωb97xd/6-31G(d) optimized structures of the three conformers of 1.
Note that the authors did not report their structures in their supporting materials(!) so I have optimized them.
Note that the authors did not report their structures in their supporting materials(!) so I have optimized them.
The structures of conformers 1a and 1b are nearly planar. MP2 predicts a non-planar rotomer of 1a, which brings the carboxyl group out of plane, to be the lowest conformation in terms of electronic energy. With the M06-2x functional, this non-planar rotomer is about isoenergetic with 1a. With all computational levels 1a is the lowest in free energy. The barrier for rotation between the non-planar rotomer and 1a is very small, and this explains why it is not observed in the supersonic expansion.
References
1) Cabezas, C.; Varela, M.; Alonso, J. L., "The Structure of the Elusive Simplest Dipeptide Gly-Gly." Angew. Chem. Int. Ed. 2017, 56, 6420-6425, DOI: 10.1002/anie.201702425.
InChIs
1: InChI=1S/C4H8N2O3/c5-1-3(7)6-2-4(8)9/h1-2,5H2,(H,6,7)(H,8,9)
InChIKey=YMAWOPBAYDPSLA-UHFFFAOYSA-N
InChIKey=YMAWOPBAYDPSLA-UHFFFAOYSA-N
 '
'This work is licensed under a Creative Commons Attribution-NoDerivs 3.0 Unported License.
No comments:
Post a Comment